Why Do Solar Panels Absorb Mostly Visible Light (Not UV or Infrared)?
Our sun emits a wide range of electromagnetic radiation, from deadly gamma rays to harmless radio waves. Still, the solar panels only absorb visible light and part of the infrared radiation to generate electricity.
Why? In this post, we’ll explore the unique science behind this.
What are Photons and How do they carry energy?
The energy travels in the form of small packets called photons. In fact, they are the smallest particles of electromagnetic energy that travel at the speed of light (3 x 108 m/s). They do not carry any charge; they are neutral. This electromagnetic energy can travel even in space with no atmosphere and produces magnetic and electric fields.

The wave propagation is perpendicular to the electric field and the magnetic field, and these 2 fields are also perpendicular to each other. Each photon coming from the sun has its own energy, which can be found as:
Energy(E) = hν
Where;
- E = energy of the Photon
- h = Plank’s constant 6.626 × 10⁻³⁴ J·s
- ν = frequency of the photon
This energy could be as high as 100keV to as low as 10-6eV.
This means that the energy from the sun does not have a one set of values,

Rather, it is coming as a band of energy, and this band of energy is called the electromagnetic spectrum.
Electromagnetic Spectrum: Wavelength, Frequency and Photon Energy
We classify photons based on the value of the energy they contain.
| Region | Wavelength Range (approx.) | Frequency Range (Hz) | Photon Energy Range (eV) |
|---|---|---|---|
| Gamma Rays | < 0.01 nm | > 3 × 10¹⁹ Hz | > 100 keV (10⁵ eV) |
| X-Rays | 0.01 nm – 10 nm | 3 × 10¹⁶ – 3 × 10¹⁹ Hz | 100 eV – 100 keV |
| Ultraviolet (UV) | 10 nm – 400 nm | 7.5 × 10¹⁴ – 3 × 10¹⁶ Hz | 3 eV – 100 eV |
| Visible Light | 400 nm – 700 nm | 4.3 × 10¹⁴ – 7.5 × 10¹⁴ Hz | 1.65 eV – 3.1 eV |
| Infrared (IR) | 700 nm – 1 mm | 3 × 10¹¹ – 4.3 × 10¹⁴ Hz | 0.001 eV – 1.65 eV |
| Microwaves | 1 mm – 1 m | 3 × 10⁸ – 3 × 10¹¹ Hz | 10⁻⁵ eV – 10⁻³ eV |
| Radio Waves | > 1 m | < 3 × 10⁸ Hz | < 10⁻⁵ eV |
- Gamma rays have the highest photon energy (> 100 keV = 10⁵ eV).
- Radio waves have the lowest photon energy (< 10⁻⁵ eV).
- Visible spectrum (what our eyes see) lies between ~1.65 eV (red) to ~3.1 eV (violet).

The Earth’s Atmosphere is selective: Not all radiation reaches solar panels
The Earth’s atmosphere acts like a natural shield against harmful high-energy radiation from the Sun. Our Earth’s atmosphere is transparent to visible light, most of the infrared radiation, some microwaves, and all radio waves. It selectively blocks Gamma, X-rays, and UV rays.

Why are UV and X-rays blocked before reaching Earth?
X-rays and Gamma rays: These are extremely energetic and harmful to living beings. Fortunately, they are almost completely absorbed by the upper atmosphere (ionosphere and thermosphere) and do not reach the Earth’s surface.
Ultraviolet (UV) rays: Only a small fraction of UV radiation reaches us. The ozone layer in the stratosphere absorbs nearly all of the Sun’s high-energy UV-C and most UV-B radiation. Only some UV-A and a little UV-B penetrate to the surface, which is why excessive exposure can still cause skin damage. Finally, visible light, most of the infrared radiation, some microwaves, and all radio waves are available for solar panels for the solar PV effect.

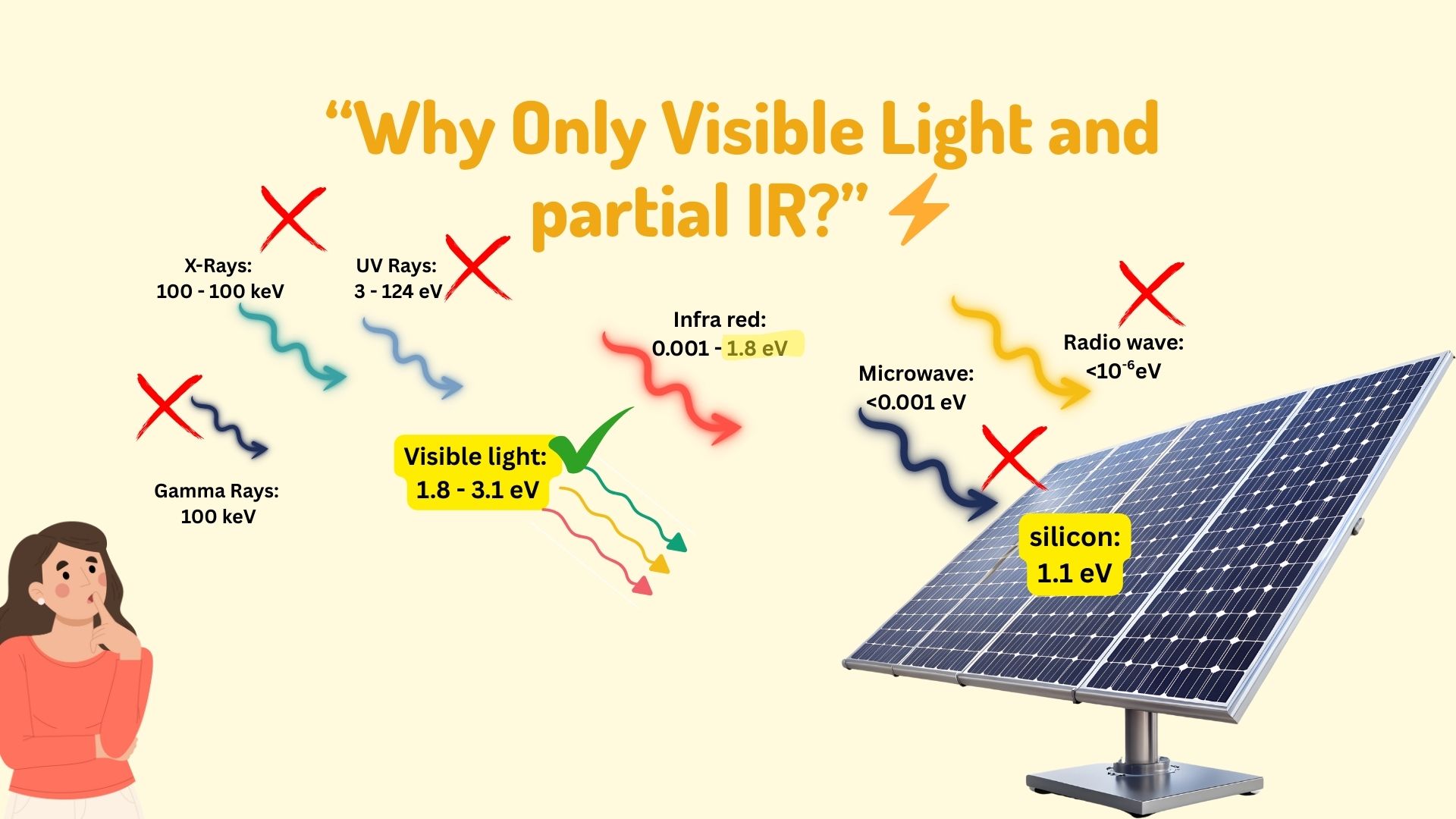
Why are only visible light and partially infrared radiation absorbed by the solar panels?
To find the answer to this question, we need to look at the solar panels. The solar panels are made of silicon.

About 90% of the solar panels in the world are made of silicon. It has a unique property of converting sunlight into electricity, called the photovoltaic effect, and is thus used in making solar cells.
What happens when light hits the solar cell?
When sunlight (photons) hits a solar cell:
- If the photon’s energy is greater than or equal to the band gap, it excites an electron → current is generated.
- If the photon’s energy is less than the band gap, it passes through without being absorbed.
- If the photon’s energy is much higher than the band gap, the extra energy is wasted as heat.
Silicon bandgap in Solar Panels
The silicon has a bandgap of 1.1eV, and this makes it ideal for converting the visible light into electricity.

The bandgap is the minimum energy required by an electron in the valence band to jump to the conduction band and become free to conduct electricity.
And this minimum energy is 1.1eV. If a photon of light has energy >=1.1eV, which the visible spectrum has, it will set the electron free and make it available for the photovoltaic effect. Anything below 1.1eV will not contribute to the PV effect and will be wasted as heat in the cell.

Therefore, it is the silicon bandgap of 1.1eV and visible light energy of 1.8eV to 3.1eV, making it an ideal match to create the photo-voltaic effect in solar panels.
Also, the part of the infrared radiation, only the near-infrared photons above 1.1 eV (λ ≤ 1100 nm) contribute; lower-energy far-infrared does not. energy is also higher than the silicon bandgap of 1.1eV. Hence, it also contributes to the PV effect, but partially.
Why is Silicon the perfect material for Solar Panels?
The natural question that arises in everybody’s mind is,” Why is 90% of the time silicon is used in making solar panels? We could have used another material.
Well, the following points justify its use:
- Silicon is the second most abundant element in Earth’s crust (after oxygen).
- This availability makes silicon-based solar panels relatively affordable compared to exotic materials.
- Silicon solar panels last 25–30 years with only slight degradation (~0.3–0.5% per year).
- Their long stability makes them the industry standard.
- Good Bandgap: Matches the Sun’s visible spectrum well.
Key Takeaway: Sunlight and Silicon’s Perfect Match
- Silicon bandgap ≈ 1.1 eV → only photons with energy ≥ 1.1 eV (λ ≤ ~1100 nm) can generate electricity.
- This means:
- Visible light (400–700 nm, 1.8–3.1 eV) → highly useful.
- Near IR (700–1100 nm, ~1.1–1.8 eV) → partially useful.
- Far IR (>1100 nm, <1.1 eV) → wasted as heat.
- UV (>3 eV) → partially absorbed, but excess energy is lost as heat.
So, the visible part of the spectrum is the most important for solar power. This is also why solar panels don’t generate much electricity on cloudy days, because visible light is reduced, and most of the infrared radiation cannot be used effectively.
“So now you know — solar panels work best with visible light because silicon’s bandgap matches perfectly with the energy of visible photons. That’s why the Sun’s light and silicon make a unique combination to power our world.”
Here are quick answers to the most common questions about why solar panels absorb visible light and not UV or infrared radiation.
FAQ: Why Solar Panels Absorb Visible Light (Not UV or Infrared)
1. Do solar panels absorb UV light?
Only partly. UV photons have excess energy, which is mostly lost as heat.
2. Why don’t solar panels use infrared (IR)?
Most IR photons have too little energy to cross silicon’s bandgap, so they pass through.
3. Why is visible light best for solar panels?
Visible photons (1.65–3.1 eV) match silicon’s 1.1 eV bandgap, making them most efficient.
4. Can solar panels capture more than visible light?
Yes. Multi-junction and thin-film cells can absorb more spectrum, but cost is higher.
5. Why is silicon used in most solar panels?
It’s abundant, affordable, durable, and perfectly suited to the Sun’s visible spectrum.
Want to Go Beyond Basics: Join the Master Class
If you’re serious about solar, check out my Solar Energy Masterclass: Basics to Profit. You’ll learn how to design, size, and calculate the payback of solar systems step by step.